Elon Musk’s Neuralink shows brain-chip patient playing online chess
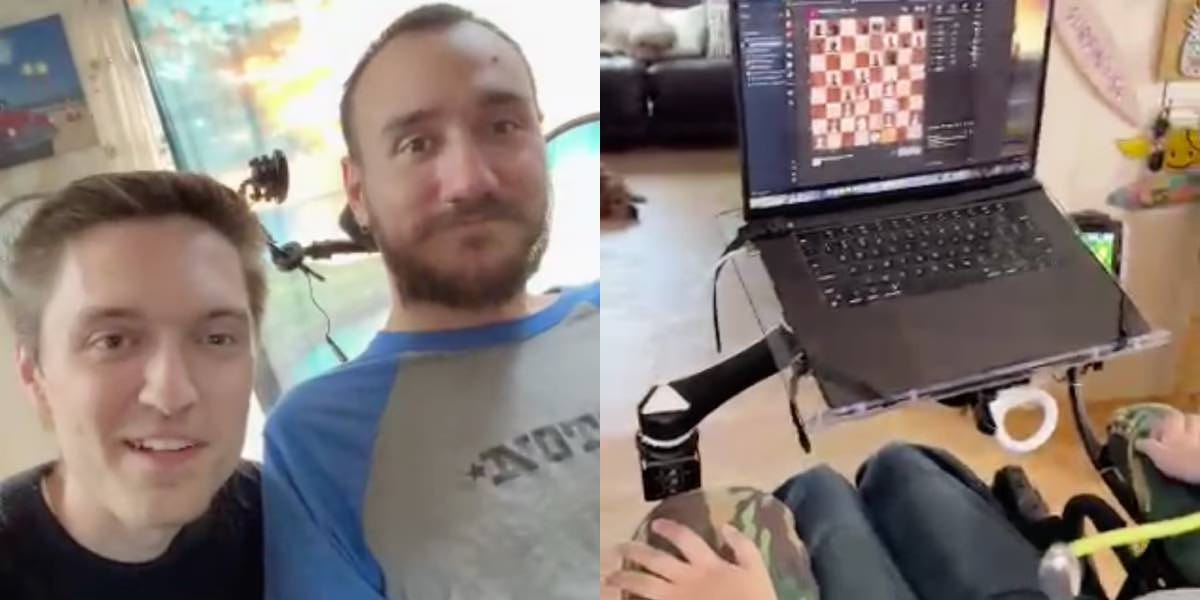
In a post on X (formerly Twitter), Neuralink, the neurotechnology startup that specializes in creating brain implants, live-streamed the first chip-implanted patient playing online chess.
Noland Arbaugh, the 29-year-old patient who was paralyzed from the shoulder down after a diving accident, was playing chess on his laptop and moving the cursor using the Neuralink device. "If you can see the cursor moving around the screen, that's all me," he said during the live stream as he moved a digital chess piece. "It's pretty cool, huh?"
"Progress is good and the patient appears to be fully recovered, with no side effects that we know of," Musk said on Feb. 19 in a Spaces on X audio chat in response to a question about the participant's condition. "The patient is able to move a mouse around the screen just by thinking," he added.
"The surgery was super easy," Arbaugh said in the video streamed by Platform X, referring to the implant procedure. “I was literally out of the hospital the next day. I don't have any side effects."
"I had basically stopped playing that game," Arbaugh said, referring to the Civilization VI game, "everyone [at Neuralink] made it possible for me to do it again."
The live stream contained relatively few details, a Neuralink engineer said in the video that more information will be released in the coming days.
Kip Ludwig, former program director for neural engineering at the US National Institutes of Health, said what Neuralink showed was not a "breakthrough".
"It's still in the early days post-implant, and there's a lot of learning needed on both the Neuralink side and the patient side to maximize the amount of control information that can be achieved," he added.
Even so, Ludwig said it was a positive development for the patient that they were able to connect to a computer in a way they couldn't before the surgery. "It's definitely a good starting point," he said.
The company received the green light from the United States Food and Drug Administration last year to proceed with an initial human trial and began seeking people with mobility issues in the fall to test the device.
Last month, Reuters reported that U.S. Food and Drug Administration inspectors found problems with record-keeping and quality controls for animal experiments at Elon Musk's Neuralink, less than a month after the startup said it approved to test its brain implants on humans. Neuralink declined to answer questions about the FDA inspection at the time.
Watch the video here.